 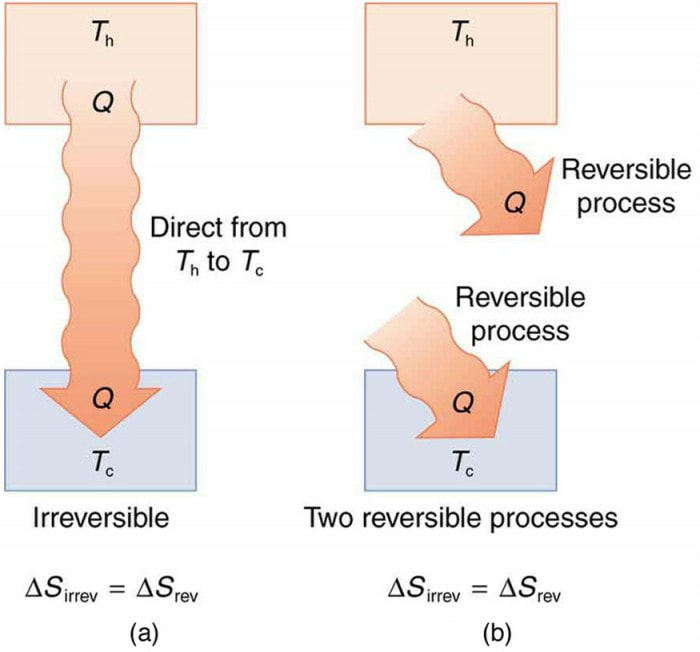
(3) The heat transfer of the irreversible process cannot be simply calculated without additional information, and it cannot be shown in the T-S diagram.Įntropy generation is another important concept in the second law of thermodynamics. 
Figure 6.5.e2 T-S diagram: the shaded area represents the heat transfer of a reversible process. 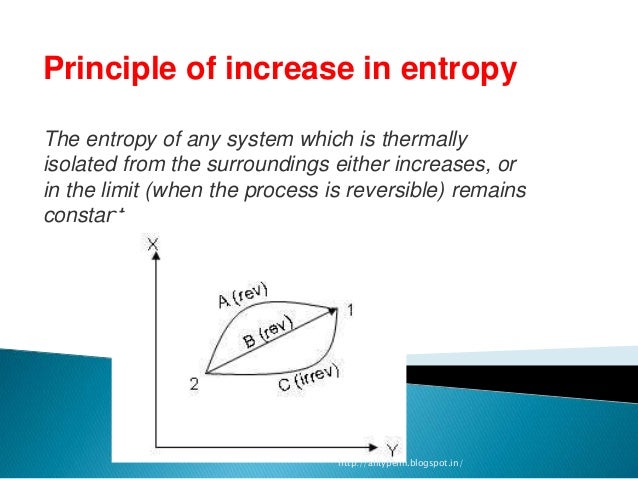
Specific entropy has a symbol s and the units are J/kg K It should be pointed out that there are other definitions of entropy but this one is the most meaningful for thermodynamics. By convention, it is often chosen as the triple-point temperature for water, which turns out to be an appropriate choice here as well. We show that pointlike defect model of glasses cannot explain the thermodynamic properties of glass formers, as for example, the excess specific heat close. This is the way entropy was developed for thermodynamics and from the above we get the definition dS dQ/T The units of entropy are hence J/K. If Suniv < 0, the process is nonspontaneous, and if Suniv 0, the system is at equilibrium. Considering it an ideal gas and a constant cp c p value. This integral can be shown graphically as the shaded area under the T-S curve of the reversible process, see Figure 6.5.e2. The specific entropy for the liquid, when assuming incompressibility, is (5) Again, the value of the integration constant T 0 plays no physical role in our application. The second law of thermodynamics states that a spontaneous process increases the entropy of the universe, Suniv > 0. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
